We are no longer accepting new cases.
When used during surgery on patients undergoing hysterectomies or myomectomies (removal of the uterus or uterine fibroids), these devices can spread a hidden and rare, yet aggressive, uterine cancer. Patients are left with a poor prognosis of recovery.
In July, the FDA convened with the Obstetrics and Gynecological Medical Devices Panel to evaluate whether or not it is safe to continue using power morcellators in gynecologic surgeries. The panel felt that, in most cases, the risks outweigh the benefits.
It recommended that doctors explain to their patients the magnitude of the risk when they agree to surgery, and said they would consider requiring “black box” warning labels — the most severe warnings the FDA has. Leading hospitals nationwide are considering banning their use altogether or restricting how the tools are used.
In addition, Johnson & Johnson, one of the manufacturers of these surgical devices, discontinued all sales of its laparoscopic power morcellators following the FDA warning. J&J products affected by this action include the Gynecare Morcellex Tissue Morcellator, the Morcellex Sigma Tissue Morcellator System, and the Gynecare X-Tract Tissue Morcellator.
W&L Attorneys Handling Power Morcellators Litigation
Melinda Davis Nokes, Associate Attorney

Melinda Davis Nokes enjoys a well-deserved reputation for ingenuity and tenacity when it comes to securing experts who can offer powerful testimony about the connection between dangerous pharmaceuticals and the injuries they can cause.
Lawrence B. Goldhirsch, Trial Counsel

Lawrence B. Goldhirsch has been with Weitz & Luxenberg since 1994 and serves as trial counsel in our Drug & Medical Devices Litigation group.
Michael E. Pederson, Associate Attorney

Michael E. Pederson fights for the rights of adults and children who become injured by pharmaceuticals and medical products purported to be safe.
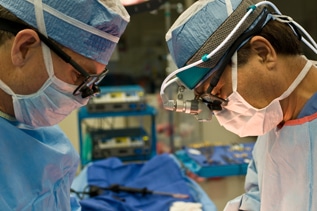
What Are Power Morcellators?
Power morcellators are tiny surgical devices with rapidly rotating blades. Often when removing the uterus or uterine fibroids, surgeons insert these devices through a small incision in the patient’s abdomen. The tiny blades then slice the tissue into small pieces that can be sucked out through the opening of a laparoscope.
Power Morcellators Can Spread Cancer
In the process of slicing up the uterus or uterine fibroid, power morcellators can spread previously undiagnosed cancerous tissues throughout the abdominal cavity. These cancerous growths are of particular concern because there is no way to distinguish them from benign ones until they are removed and evaluated.
In addition, typical symptoms may simply be signs of any number of non-life-threatening conditions.
Common symptoms may include:
- Abnormal vaginal bleeding, spotting or discharge
- Pelvic pain
- Palpable mass
Prognosis for an Aggressive Uterine Sarcoma Is Poor
There are four main types of uterine and endometrial cancers:
- Leiomyosarcoma — cancer that begins in smooth muscle cells
- Endometrial stromal sarcoma — cancer that begins in connective tissue cells
- Endometrial adenocarcinoma — the most common type of endometrial cancer
- Uterine carcinosarcoma — cancers that begin in the cells that line the uterus
Of these, leiomyosarcoma is particularly aggressive and alarming. Endometrial stromal sarcoma is generally slow growing.
If leiomyosarcoma is left undisturbed and intact in its original location when the patient is diagnosed, the five-year survival rate is roughly 63 percent. If the cancer has been disturbed and spread throughout the abdominal cavity by power morcellators, depending on the severity, the five-year survival rate drops from 36 to 14 percent.
Complications of Power Morcellators in Gynecologic Surgery
There are several significant complications that may arise when power morcellators are used in surgical procedures:
- Spread of uterine leiomyosarcoma, a rare but aggressive cancer
- Spread of endometrial stromal sarcoma
- Spread of endometrial adenocarcinoma
- Spread of uterine carcinosarcoma
- Spread of benign tissue, which can lead to pain, infection or bowel obstruction
- Injury to surrounding organs, including the bowel, bladder, pancreas and spleen
- Parasitic growth of retained tissue
Women Suffering from Complications May Be Entitled to Compensation
If you have recently undergone a hysterectomy or myomectomy to remove uterine fibroids, you are not alone. Annually, more than 500,000 women may be planning to undergo this type of surgery.
Unfortunately, for a small percentage of women, the complications following such a surgery involving power morcellators can be life threatening.
If you are experiencing severe symptoms or have been diagnosed with uterine sarcoma as a result, you may be entitled to compensation.