Replacement surgeries most commonly involve the hip or knee, although most joints in the body can be replaced including shoulders, wrists and elbows.
For hip replacement alone, more than 450,000 surgeries are performed annually. Ideally, patients today should expect a hip or knee replacement to last for 20 or more years.
Complications, however, do occur. In fact, for joint replacement in general, the percentage of people experiencing complications may be as much as 40 percent.
For a free consultation and more information about your legal options, please contact us today.
Get a Free Case ReviewWhat Is Joint Arthroplasty Surgery
Joint replacement surgery, also known as arthroplasty, involves removing the worn or damaged cartilage from both sides of the joint, followed by resurfacing the joint with a replacement implant. The implant is designed to mimic a normal joint and should allow patients to resume a high degree of normal activity within a few months.
Some pain following surgery is to be expected. However, if it is severe, or is manageable at first but worsens over time, that is a concern. It could mean that the joint replacement is failing.”
New Materials and Methods Are Not Necessarily Better
Historically, the use of hip implants was limited to patients who were older. These patients were less active, had suffered a hip fracture or experienced severe arthritic hip conditions.
Currently, orthopedic surgeons have been offering hip replacement options to patients as young as 55, perhaps younger. Implants are being made of a variety of materials, including combinations of metal, polyethylene and ceramic.
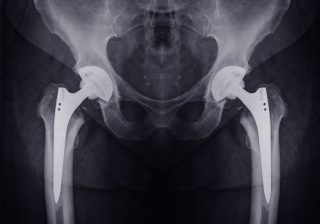
Herein lies good news and bad. With the demand for products and devices suitable for younger, sometimes far more active patients, manufacturers have been aiming to create stronger, more durable, more flexible designs. In some cases, this has led to defective products that have been inadequately tested and researched.
Complications
There are almost as many complications as there are joints in the body.
Many of them are related to having any type of surgery:
- Infection
- Inflammation
- Blood Clots
- Pain
- Dislocation
- Numbness
- Bleeding
- Nerve and blood vessel damage
- General hypersensitivity reaction (skin rash)
- Soft-tissue damage
Other have more to do with joint surgery, specifically:
- Local tissue necrosis, or tissue death
- Deterioration of the bone around the implant
- Metallosis due to the buildup of metallic debris in the soft tissue surrounding the joint
- Bone growth beyond the normal edges of bone
- General wear and tear, requiring revision surgery
Other complications have definitive mechanical implications:
- Loosening
- Fractures
- Noise (popping, grinding, clicking or squeaking of your hip)
- Change in ability to walk
- Device failure
- Prosthetic breakage
- Leg-length inequality
Determining if Your Implant is Defective
In many cases, you may not be aware what type of implant you received. A good lawyer can determine if you have been subjected to a defective device. With your permission, an experienced lawyer can get access to your records and help you understand your options.
Selecting the Attorney to Best Represent You
Choosing the right attorney may feel overwhelming when you are suffering from a joint replacement failure. To select the firm that will best represent you, you need to ask the right questions. What experience can this firm offer me? What is this firm’s philosophy toward working with its clients?
Weitz & Luxenberg stands out for its success in winning complicated medical lawsuits. We represent tens of thousands of individuals, consistently offering superior results. You can be assured that if you select Weitz & Luxenberg, we will fight with you and for you every step of the way.
We would feel privileged to assist you. For a free consultation and more information about your legal options, please contact us today.
(917) LAWYERSHow Weitz & Luxenberg Can Help
As a nationally recognized personal injury law firm, Weitz & Luxenberg is committed to helping clients win cases. For almost 40 years, we have dedicated ourselves to holding irresponsible practitioners accountable, and we have won $26 billion for our clients.
We would feel privileged to assist you. For a free consultation and more information about your legal options, please call us at (917) LAWYERS. If you prefer, you can complete our form, and our client relations representative will contact you shortly.